Vioxx and COX2 inhibitors
Much has been said recently about the effects of COX2 inhibitors on the heart, in particular coronary artery disease. COX2 is the abbrevation for selective cyclo-oxygenase2 inhibitors, the receptors that help to lessen pain. Examples are drugs like VIOXX, CELEBREX, ARCOXIA etc.
Before the advent of COX2 inhibitors, we had non-selective COX inhibitors (sometimes called COX 1 inhibitors) like NSAIDs (voltaren, cataflam, mobic, aspirin, naproxen, etc.) The problems with COX 1 inhibitors is that, besides alleviating pain, they also cause gastric problems as there are COX 1 receptors in the stomach, and blocking them make them prone to gastric epithelial denudation and the accompanying gastric upset and perhaps gastric bleeding too. In an attempt to lessen the gastric side-effects, COX2 inhibitors were invented.
We became interested in this problem back in 2002 (before the Vioxx controversy exploded on the front page) when the Novartis asked me to look into the problem of "COX2 and Atherothrombosis". Their interest may have been prompted by a paper on CLASS study, that was published in BMJ in June 2002.
Having looked into the issue, I agreed with Novartis that COX2 is potentially harmful for patients suffering from CAD. There were obvious pathogenetic mechanisms to explain this harm. When I examined the CLASS study and also the VIGOR study, I was very surprised that the FDA approved the COX2 as there were inconsistencies in the data pesented. When I mentioned these in the talks that I subsequently gave on behalf of Novartis entitled "COX2 and atherothrombosis", the audience (remember, this was in yr 2002) were quite sceptical.
You may wish to know that the VIGOR study compared the effects on the stomach of ROFECOXIB (Vioxx) against NAPROSYN. CVS risk was a post-hoc analysis which showed an increase incidence of major adverse cardiac events with the use of rofecoxib. The investigators concluded that this could be due to the cardio-protective effects of naprosyn and the FDA did not pursue the matter.
In the CLASS study, the investigators compared the gastro-toxic effects of Celecoxib against a Ibuprofen and Diclofenac. Again, it was not a CVS effects study, and the CVS effects was very much a post-hoc subgroup analysis. In this study, there was no increased incidence of major adverse cardiac events in the celecoxib group. This may be partly because many of the subjects on celecoxib, were also taking aspirin as prevention against CAD. One way of avoiding major adverse cardiac events with the use of COX2 inhibitors is to use it with a small dose of aspirin.
The Theory on COX2 and the heart
Firstly, it is important to note that many tissues in the body has cells that expresses the COX receptors. These tissue include gastric mucosa, fibrous tissues in the joints, and more importantly, platelets and endothelial cells of the artery wall. These tissue have many kinds of COX receptors. Of importance are the COX1 receptors (which are non-specific) and the COX2 receptors which are specific and selective.
These receptors are stimulated by pain and inflammation. Generally speaking, stimulation of the COX receptors produces pain and inhibition of these receptors reduces pain. Although these actions are important in joint pains and inflammation, other effects are more important as far as artherothrombosis is concern.
Almost as if nature must maintain everything in balance, when the COX1 receptors in the platelets are stimulated, it produces thromboxane A2 which causes platelet aggregation and vasoconstriction. When the COX1 receptors in the endothelial cells are stimulated, it produces prostacyclin which causes vasodilatation and also inhibit platelet aggregation. The crucial factor is that the nucleated endothelial cells has COX2 receptors whereas the non-nucleated platelets do not have COX2 receptors. Therefore during episodes of inflammation, platelets aggregate and vasoconstriction is triggered. But those actions are opposed by the prostacyclin from the endothelial cells with counteraction of platelet non-aggregation and vasodilation.
A "yin-yang" like hemostatic balance is maintained and the artery is largely un-affected. Now when we use NSAIDs as anti-inflammatory agents, NSAIDs block the non-selective COX1 receptors equally on the platelets and endothelial cells, and so the hemostasis is balance. But with the use of COX2 agents, they can block COX1 receptors in platelets and endothelial cells (thats okay) but they block selective COX2 receptors, located only in the endothelial cells, thereby diminishing the prostacyclin, so vital for vaso-dilatation and platelet non-aggregation.
The following four slides may somewhat provide a better picture of what's happening (please email me for a copy of the slides).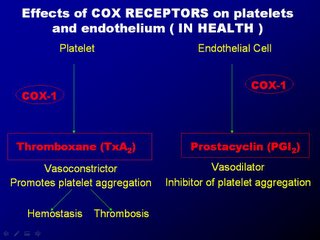
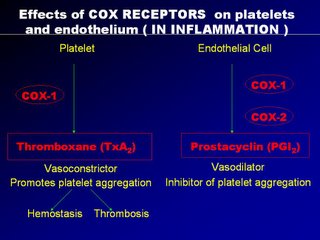
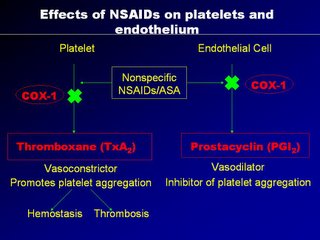
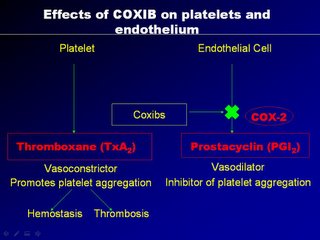
The result is unopposed vasoconstriction and platelet aggregation. These effects are potentially deletarious in anyone with CAD (known or silent). It's good to note that osteoarthritis affects mainly the elderly, a population at high risk of CAD. Basically, COX2 inhibition is theorectically pro-atherogenic. These was elegently demonstrated in a study by scientist Dr Herman in an study published in Circ. Vol 104 page 820, yr 2001 entitled "Effects of selective cyclooxygenase 2 inhibition on vascular responses and thrombosis in canine coronary arteries".
What was most upsetting is that FDA probably knew about it and did nothing about it. And I quote from the editorial on the CLASS study published in BMJ 1st June 2001.
"Publishing and distributing overoptimistic short term data using post hoc changes to the protocol, while omitting disappointing long term data of two trials, which involved large numbers of volunteers, is misleading. While some of the problems related to CLASS were partially covered in the news sections of BMJ 11 and other journals, it was not emphasised how flawed the trial actually was, and how inadequate the authors' justifications. Consequently, CLASS may still be relied on by many physicians without reference to these flaws. In our experience most still believe the findings published originally. For example, most of 58 physicians attending an osteoarthritis workshop in Berne, Switzerland, in December 2001 had not realised that CLASS was seriously biased."
I therefore believe that on this issue, vioxx has a case to answer.
No comments:
Post a Comment